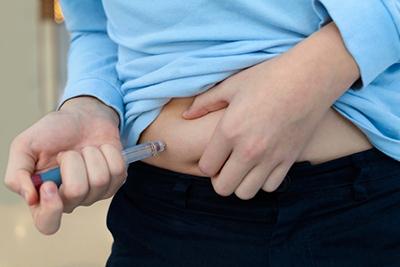
-
Benefits of Mounjaro
posted: Dec. 14, 2023.

-
CLEAR wisdom and why is this being presented at the March 18th Late breaking Clinical Trial session in New Orleans?
posted: Jan. 30, 2019.
What is CLEAR Wisdom? It sounds like something from your doctor of philosophy and your brain, but it’s really about a potentially valuable new treatment for your heart. If you Read more -
Credence - Invokana True Breakthrough
posted: Jan. 14, 2019.
True Breakthroughs But when will the CREDENCE results on Invokana be published? You may remember that the CREDENCE study was stopped early July 16, 2018 --the “Phase 3 CREDENCE (Canagliflozin and Read more -
Looking beyond lipids, is there swelling evidence?
posted: Nov. 09, 2018.
By James Joseph Rybacki, Pharm.D. 1/8/19 Even when patients are treated to blood pressure and cholesterol goals, they still have heart attacks and some of them die. This sobering fact has Read more -
Xarelto gains a new CAD/PAD indication, what now?
posted: Nov. 09, 2018.
Coming soon. Read more -
Exercise (physical activity guidelines) from HHS—did they hit the mark?
posted: Nov. 09, 2018.
Coming soon. Read more -
REDUCE-IT reduced MACE by 25%, does it validate Vascepa?
posted: Nov. 09, 2018.
The American Heart Association (AHA) Scientific Sessions November 10-12th in Chicago will have great science as always, but may stand out as having the most significant impact on cholesterol management, Read more -
Will November 10th late breakers from the American Heart meeting validate Vascepa?
posted: Nov. 09, 2018.
Coming soon. Read more -
Labeling changes for Vascepa? Who will now pay for the drug?
posted: Nov. 09, 2018.
Have you heard about the REDUCE-IT study? For those attending the American Heart Association meeting in Chicago By James Joseph Rybacki, Pharm.D. 1/8/19 On November 10-14 2018, you could have heard a Read more -
Guiding gathering for new cholesterol guidelines, what’s the threshold?
posted: Nov. 09, 2018.
Greeting new Guidelines or should we embrace them and quickly integrate them into practice? By James Joseph Rybacki, Pharm.D. 1/8/19 You may have noticed that guidelines are updated about every 2-3 years. Read more -
Will Andexxa answer crucial reversal questions?
posted: Nov. 09, 2018.
Coming soon. Read more -
Can canakinumab solve the inflammation flame?
posted: Nov. 09, 2018.
Coming soon. Read more -
Why can’t my Mom get a PCSK9?
posted: Nov. 09, 2018.
What on earth is the controversy over PCSK9 medicines for cholesterol.?? By James Joseph Rybacki, Pharm.D. 1/8/19 Hmmmm, it comes down to money, or it used to, or, it still does. Let’s Read more -
Will 2018 AHA be an aha moment for cardiology?
posted: Nov. 09, 2018.
Coming soon. Read more -
Twitter and medicine, is it a righteous mix?
posted: Nov. 09, 2018.
Did you think that Twitter would become mainstream medicine? By James Joseph Rybacki, Pharm.D. 1/8/19 I wasn’t so sure when I decided to call myself Rybackiknows. Sounded pompous at first, then had Read more -
Will 2018 AHA be an aha moment for cardiology?
posted: Nov. 09, 2018.
Coming soon. Read more
